CHARLOTTE, N.C. — Photos often tell the story of our lives. Happy beginnings, trips where smiles and memories are made, momentous occasions to celebrate and mark milestones.
But sometimes, life hits the pause button on happiness for no good reason at all.
Lisa Stockman-Mauriello's heartbreaking story centers around her struggle for access to a drug that will be a game-changer in her fight live longer. Just long enough to see her kids graduate in a few months.
Stockman-Mauriello is a University of North Carolina alumna and lived in the Charlotte area for years before relocating to New Jersey.
“Moms aren’t supposed to be sick” Lisa said in a soft voice.
"Moms aren't supposed to be sick," Bob Mauriello, Lisa's husband, agreed.
Lisa is very sick, and Bob is slowly becoming her voice. Lisa was diagnosed with Bulbar ALS, a very rare kind of ALS that progresses rapidly because of the mutation of the SOD1 gene. It's quickly robbing her of the ability to speak, the ability to move and soon, her ability to live.
It is, in a word, heartbreaking.


A new drug, now in clinical trials, might help Lisa live longer. She's hopeful the drug could help her get what any mother would want.
"Long enough to see our sons graduate in the spring," Lisa said. "One from college, one from high school, and one from the eighth grade."
Lisa’s physician, Dr. Neil Shneider, a neurologist and Director of the Eleanor and Lou Gehrig ALS Center at Columbia University, said this drug may indeed help her.
“I think there is evidence to suggest that this therapeutic would be helpful to her, and you know, in ALS, we don’t have a lot of therapeutic options, we are very limited as to what we can offer our patients, so I feel this is her best chance, for a therapeutic that could make a meaningful difference in her disease,” Dr. Shneider saad.
This is where things get complicated and emotional. Biogen is the innovator of the new drug Tofersen, It's the drug that could help Lisa and others like her live longer.


"It's in phase three clinical trials," Bob Mauriello said. "We'd be happy for Lisa to participate in the trial but it closed at the end of the year, just weeks before she was diagnosed."
Despite the red tape, Lisa, Bob and her family officially petitioned Biogen asking for this drug. So has Dr. Shneider, as well as tens of thousands of supporters who signed a petition asking Biogen to let her have Tofersen under compassionate use.
But the answer came late last week in a letter. Biogen said no, denying Lisa's request.
“Let me begin by expressing my personal, and our team’s collective empathy upon learning of your ALS diagnosis. We can only imagine the magnitude of the devastation that you and your family must be experiencing. Since we learned of your request for early access to Tofersen, which is still being tested in a clinical study, we’ve rapidly brought together our medical, research, regulatory and senior leadership colleagues to thoroughly evaluate and challenge our own policies in light of the situation for you and other people with ALS. We have also been in touch with your physician since we received your request.
Obtaining approval for a new drug from regulatory authorities around the world is the fastest way to help the largest number of people with a specific disease and requires the completion of clinical testing. Until efficacy and safety are determined, we must act with the interests of all patients in mind. Providing individual access to Tofersen at this time could jeopardize access to Tofersen for hundreds of SOD1-ALS patients by impeding our ability to complete the study that will determine whether Tofersen is efficacious and safe and to seek subsequent regulatory approvals as quickly as possible.
Our study has completed enrollment and data results are expected in the second half of this year. I want to assure you that we are working as fast as we can. We are preparing to open an Early Access Program soon after there are no more patients in our study who are on placebo and Tofersen has shown sufficient efficacy and safety to represent a positive benefit-risk for ALS patients” wrote Dr. Maha Radhakrishnan, MD, the Chief Medical Officer, Biogen.
Lisa meets a lot of the criteria for what’s called “expanded use” under FDA guidelines, meaning she could get the drug for “compassionate use” if Biogen signed off on it. Lisa, and her family, and the thousands supporting them, are hoping Biogen will reconsider. More than 60,000 people have signed this online petition to Biogen.
The clock is ticking.
At the very least, Lisa and Bob want this conversation about compassionate use for all patients, with any disease, to just get started.
“Yeah, very, very frustrating” Lisa mouthed softly.

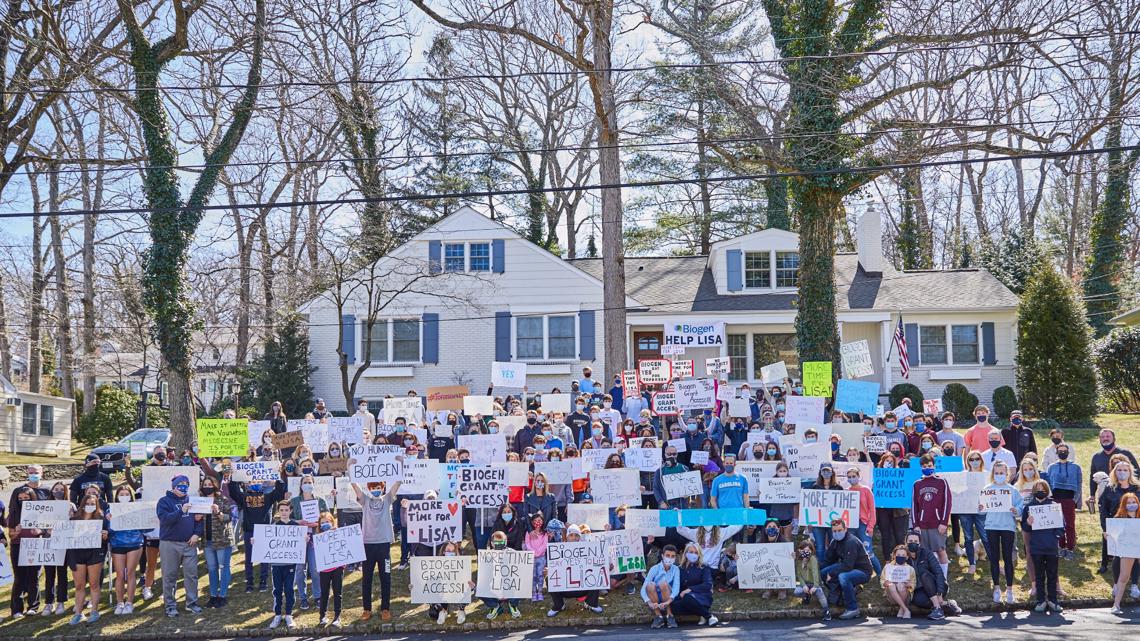
Lisa and Bob will keep fighting, and she will remain a doting mom to her three sons for as long as she can because that’s what moms do.
Biogen responded to WCNC with this statement about the denial of Tofersen for Lisa.
“We brought together people across the organization with expertise in bioethics, medicine, research and regulatory guidelines as well as our most senior leadership to challenge our own policies in light of the situation for Lisa and other people with ALS. We share the urgent desire to bring new medicines to people suffering from ALS. Obtaining approval for a new drug from regulatory authorities around the world is the fastest way to help the largest number of people with a specific disease.
Our Phase 3 placebo-controlled study of tofersen, VALOR, is designed to investigate the safety and efficacy of tofersen in people living with SOD1-ALS. The study has completed enrollment and data are expected in the second half of 2021.
We are working as fast as we can and preparing to open an Early Access Program after VALOR patients are no longer randomized to placebo and if VALOR data show that tofersen is safe and effective.
Providing individual access to tofersen at this time could jeopardize access to tofersen for hundreds of SOD1-ALS patients by impeding our ability to complete the study and seek subsequent regulatory approvals” wrote a spokesperson from Biogen’s Public Affairs department.


